RPE Development and Regeneration
Previous work in our laboratory focused on identifying novel regulators of RPE development and pigmentation, and generating zebrafish models of ocular and oculocutaneous albinism. From these studies, we identified a role for the vacuolar ATPase complex during RPE pigmentation (Nuckels et al., 2009; Shine et al., 2014), and a role for de novo purine synthesis during pigmentation, wherein a GTP-dependent pathway modulates pigment synthesis (Ng et al., 2009). Recently, we identified a zebrafish model of Hermansky-Pudlak Syndrome Type 5, and a role for the N-terminal WD40 repeat of the hps5 protein in stabilizing its interactions with hps6 in the BLOC2 complex, a well-known regulator of melanosome formation (Daly et al., 2013). From our forward genetic screen, we have identified a mutation in nsfb that results in pigmentation defects and we are currently characterizing defects in this mutant to determine how nsfb functions during melanosome formation. Recently, we have initiated a series of studies to examine RPE regeneration. Finally, we are collaborating with Brian Brooks, M.D. Ph.D. at the National Eye Institute to develop zebrafish ocular and oculocutaneous albinism models for use as a screening tool for pharmacological therapies that lead to increased pigment synthesis.
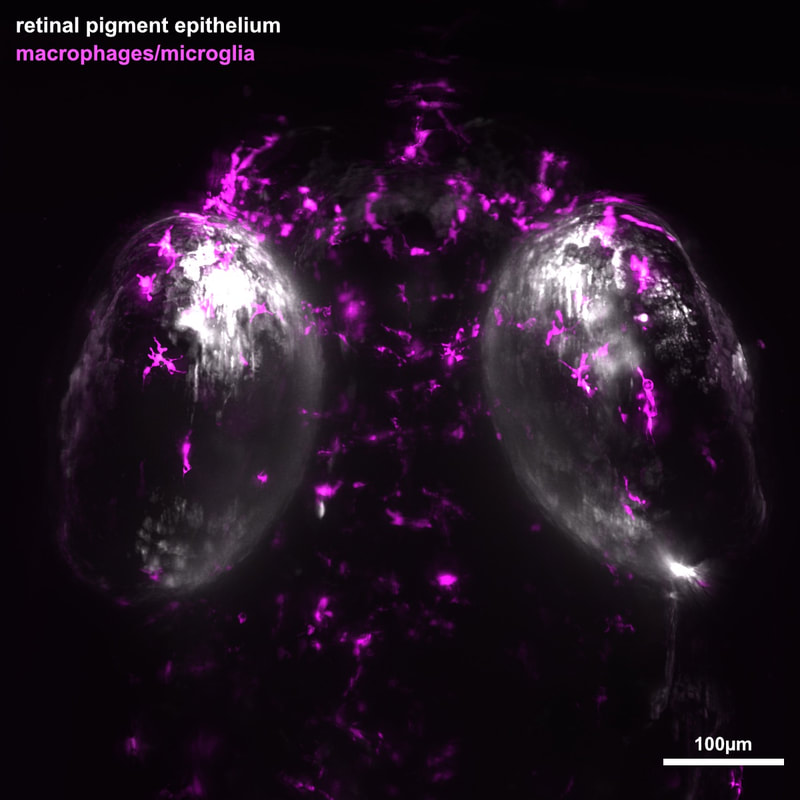
Diseases resulting in degeneration of the retinal pigment epithelium (RPE) are among the leading causes of blindness worldwide, and no therapies exist that can replace RPE or restore lost vision. Age-related macular degeneration (AMD) is one such disease, and is the third leading cause of blindness in the world. Transplantation of stem-cell derived RPE has emerged as a possibility for treating AMD and clinical trials are underway. One intriguing alternative approach to treat AMD and other diseases that affect the RPE is to develop therapies focused on stimulating endogenous RPE regeneration. Indeed, strategies targeting endogenous retinal regeneration are gaining traction as potential therapeutic approaches to treat a number of retinal degenerations. For this to become possible for the RPE, we must first gain a deeper understanding of the mechanisms underlying RPE regeneration. With this in mind we developed a transgenic zebrafish ablation paradigm to ablate large swathes of RPE. Using this model, we discovered that the larval and adult RPE regenerates after severe injury (Hanovice et al., 2019). Our results demonstrate that RPE regeneration involves a robust proliferative response during which proliferative cells move to the injury site and differentiate into RPE and that the source of regenerated cells is likely uninjured peripheral RPE. Current studies are utilizing lineage tracing to unambiguously identify the source of regenerated RPE tissue. We have also identified a role for the immune response in RPE regeneration. Our most recent results show that both inflammation and specific innate leukocytes (macrophages and microglia cells) are required for RPE regeneration to proceed normally (Leach et al., 2021). We have performed two large RNA-sequencing screens over the course of RPE regeneration in both the RPE and relevant leukocytes and are now focusing on specific cytokines, proteases, and other signaling pathways that may be critical for RPE regeneration.